Share this project
Fabrication and Mechanical Properties of Supercharged Polypeptides based Biomaterials
Summary
Bioinspired mechanical materials have been designed and fabricated for a wide range of applications. In Chapter 1, protein-based mechanical biomaterials including adhesives and fibers are reviewed. In the first part, biological adhesives inspired by mussel and sandcastle worm are described. Genetic engineering and chemical synthesis are the two methods for the fabrication of protein-based adhesives. By molecular cloning and recombinant protein expression, the mechanical behavior of protein adhesives can be exquisitely controlled over the sequence and molecular weight. Moreover, their polypeptide nature renders them biodegradable and non-toxic. In the second part, mechanically strong protein fibers have been reviewed. Bioinspired fibers from spiders and silkworms have attracted increasing attention due to their lightweight and extraordinary mechanical properties, i.e., high tensile strength and outstanding extensibility. So far, artificial protein-based fibers have been produced mainly by two approaches: chemical regeneration and genetic engineering. The extraordinary mechanical properties, biocompatibility, and biodegradability offer the opportunity to apply such protein fibers in fabrics, tissue engineering, and drug delivery.
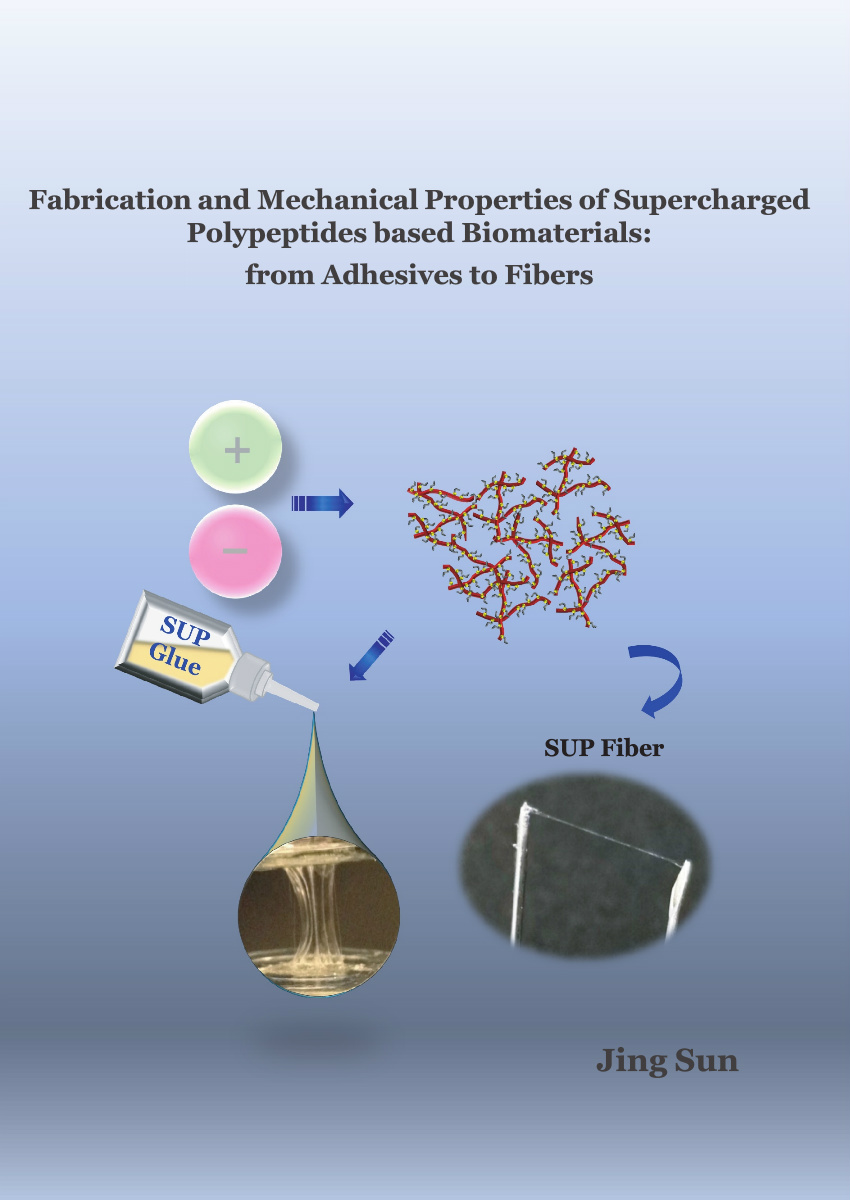
In Chapter 2, we developed a strong biological adhesive with biocompatible and biodegradable properties. The supercharged unfolded protein (SUP) glues were fabricated by employing electrostatic interaction between highly charged cationic polypeptides (cationic SUPs) and the negatively charged surfactant SDBS, followed by one-step precipitation and final lyophilization. The SUP adhesives showed significant bulk adhesion strength on various substrates, which is comparable to commercial cyanoacrylates named superglue. In addition, strong underwater adhesion was realized by this system. As an outstanding feature of this novel class of glue, during the adhesion process, covalent bond formation was avoided. It was shown through NMR spectroscopy and computational simulations that π-stacking and cation-π interactions play a critical role for strong adhesion. Moreover, the electrostatic interactions between SUPs and SDBS surfactants, van der Waals forces, and hydrogen bonds contribute to the high adhesion strength of SUP glues.
After demonstrating the adhesion ability of SUP glues, the biomedical application of this novel class of materials was investigated in Chapter 3. Firstly, the non-toxicity of the SUP glues was confirmed by high cell viability of HeLa cells in the SUP-SDBS complex. The high survival rate of mice mesenchymal stem cells (D1 cells) in 3D culturing conditions with SUP glue also indicated the safety of the adhesive system. Next, the tests on skin and eyelid adhesion of SUP glue revealed its promising properties for biomedical applications. Subsequently, the hemostatic and wound healing properties of SUP glues were investigated in vivo. The bleeding wound on rat skin and liver was sealed after treated with SUP glues, demonstrating fast hemostatic properties. Moreover, in stark contrast to suture closure and commercial chemical adhesives, fast wound healing was detected for the SUP glue, indicating the capacity of SUP glues for regenerating skin. Histological experiments were utilized to analyze the regeneration of healed skin tissue. When compared with other treatment groups, the group treated with SUP glue showed a significant formation of new blood vessels, abundant follicle formation, sebaceous glands, and more collagen deposition. Additionally, immunofluorescence analysis of the wounds treated with SUP glue did not show any signs of inflammation, which revealed the efficacy of SUP glue to prevent injury associated inflammation.
To broaden the scope of SUP glues, Chapter 4 focuses on the preparation of SUP glues with different surfactants using the same manufacturing protocol as described in Chapter 2. Firstly, the SUP glues were fabricated by employing electrostatic interaction between SUPs and relevant opposite charged DOPA or azobenzene-based surfactants. The SUP glues rendered robust adhesion both in dry and humid conditions. Particularly, in the case of the SUP-DOPA glue, bonding strength reached up to 13.51 MPa on steel because of the formation of coordination bonds between Fe3+ ions and catechol units. In this case, the adhesion is even higher than that of superglue (cyanoacrylate, 12.06 MPa). Furthermore, wound healing experiments and cytotoxicity tests proved that SUP glues exhibit good biocompatibility, low toxicity, and anti-inflammation ability. All results indicate that SUP glue can be used as a promising biomaterial for wound healing and tissue engineering.
Aside from the SUP adhesives, in Chapter 5, we report the first example for the mechanical modulation of SUP fibers reversibly in bulk state by light. We first prepared one new type of bioengineered protein fiber by employing electrostatic interactions between SUP and azobenzene (Azo) based surfactants. The macroscopic tensile tests, as well as nano-scale AFM measurements, demonstrated that the photo-isomerization of Azo moieties from the E- to the Z-form alters the tensile strength, stiffness, and toughness of the fibers reversibly. Especially, the increased cation-π interactions of the uncomplexed lysine moieties in the SUPs and the phenyl groups in Z-form of Azo leads to a ~ 2-fold increase in the fiber’s mechanics. The outstanding mechanical properties open a pathway towards the development of SUP-Azo fibers as stimuli-responsive bracing biomaterials.
See also these dissertations


Structure-Preserving Data-Driven Methods for Modeling Turbulent Flows


Molecular insights into the role of VRS5 in tillering and lateral spikelet development in barley


Gamma Knife Radiosurgery for Skull Base Tumors


Reimagining petrochemical clusters by defossilising chemical building blocks


Microbial stabilization and protein functionality of plant-based liquids using pulsed electric fields
We print for the following universities